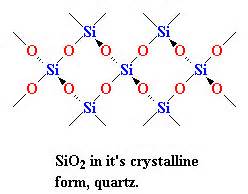
Intermolecular forces that act on SiO2
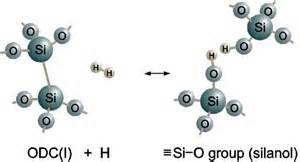
The first intermolecular force that acts on Quartz is Dispersion. Dispersion is a weak bond that occurs between ALL molecules. There is no specific rule for a molecule to acted on by the force Dispersion. So for example, if a water molecule and a quartz molecule were to bond, they could be bonded together by Dispersion. One of the Oxygen molecules from SiO2 could attach to the Hydrogen molecule from Water. This also ties into the next intermolecular force that acts on SiO2 and that is Hydrogen Bonding. Hydrogen Bonding occurs in and molecules when a hydrogen atom from one molecule bonds to either an oxygen or a nitrogen molecule from another molecule. Hydrogen usually has a positive charge while the Oxygen or Nitrogen is usually negative. In the case of SiO2, you can see how Hydrogen bonding takes place if it was bonded to Water. Again, just like with dispersion, the hydrogen from H2O, will attach to the Oxygen from the SiO2. In the picture below, you can see how Hydrogen is bonding to the Oxygen from quartz. Even though it is not as strong as covalent bonds, unlike dispersion, Hydrogen Bonding is a very strong force that acts on two molecules.