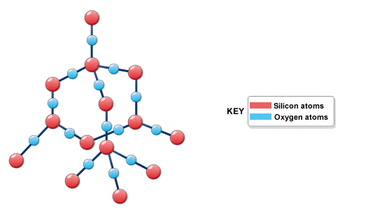
Just like in the previous picture, we can see a diagram of the structure of SiO2. Again, we can see how the molecules combine to form a network of repeating hexagons. Even though it is not shown in the picture, the angles between one Si atom and one O atom are 110 degrees. Being that when the molecules are connected, one Si is being bonded by four Oxygen molecules. An explanation to how Si and O are bonded is shown in the YouTube video below.
